by Timothy J. Sellati, Ph.D., Chief Scientific Officer, GLA
Key protein identified in study as important in defense against Lyme.
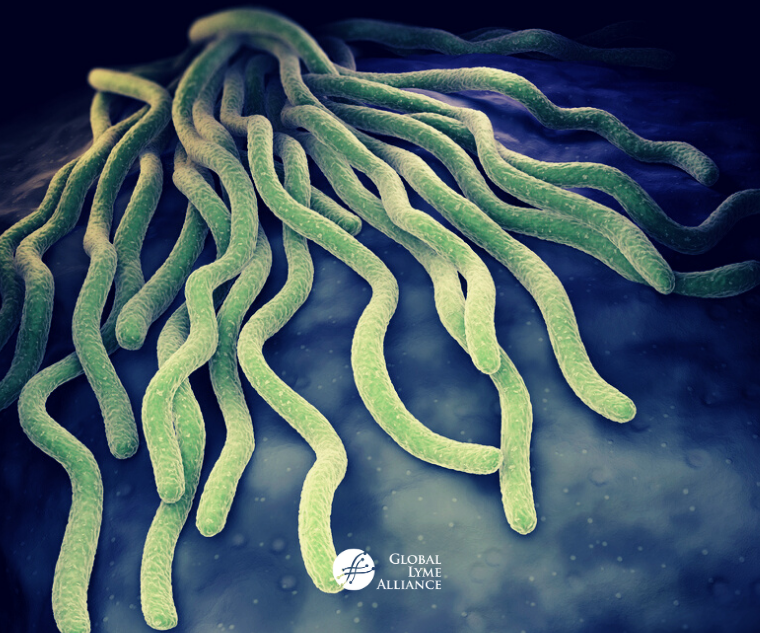
Lyme disease is the most prevalent and rapidly expanding vector-borne illness in North America.1,2 Caused by the bacterial spirochete Borrelia burgdorferi, the disease starts with a tick bite that often, but not always, leads to a classic bull’s-eye skin rash and inflammation principally impacting the joints, heart, and central and peripheral nervous systems. A new study published in PLoS Pathogens titled “Human secretome library screen reveals a role for Peptidoglycan Recognition Protein 1 in Lyme borreliosis” sheds light on how recognition of B. burgdorferi can damage our bodies in such diverse and profound ways.3
Spirochetes rely on different strategies to evade detection and persist in the human host. Many of these strategies involve binding of bacterial proteins to host proteins on the surface of immune cells responsible for defense against invading pathogens. Using a novel yeast display screening method, Dr Akash Gupta and colleagues in the laboratory of Erol Fikrig (Yale University) probed over 1,000 human immune proteins against several isolates of Borrelia to uncover biologically relevant interactions for the Lyme disease pathogen. Peptidoglycan Recognition Protein 1 (PGLYRP1) was identified as a key innate immune protein important in defense against the Lyme spirochete.
PGLYRP1 acts like an early warning signal to the immune system. When this signal is absent due to genetic mutation in mice, B. burgdorferi-infected animals had much higher levels of spirochetes in their bodies (especially in hearts and joints) and showed signs of immune system dysfunction. This finding raises the intriguing question of whether a weak signal or the complete absence of the signal in some individuals might predispose them to develop more severe and persistent Lyme symptoms. Consistent with this possibility was the finding that mice lacking PGLYRP1 also exhibited a hyperinflammatory profile. The profile was similar to that described by Drs. John Aucott and Mark Soloski (Johns Hopkins University, former GLA-funded investigator and GLA SAB member, respectively) in acute Lyme patients.4 Interestingly, the same profile also was found by Dr. Klemen Strle (current GLA-funded investigator) in mice and patients suffering from antibiotic-refractory Lyme arthritis5.
On the flip side, according to Dr Fikrig, “Stimulating the ability of people to make more of this protein could help fight infection.” With this idea in mind, researchers are investigating whether people with higher levels of PGLYRP1 may be less susceptible to infection by B. burgdorferi. If proven true, the present study suggests this might reflect the ability of PGLYRP1 to interact with B. burgdorferi peptidoglycan (PG). Among the many spirochete components that contribute to disease, it has recently been shown that B. burgdorferi sheds PG during growth, and this activity may be associated with inflammation during infection.6 PG is a structural scaffold that essentially holds a bacterium’s proteins in place as well as holds the spirochete together.
the $64,000 question is whether this killing effect can occur in a patient and, if so, could this lead to a novel approach to treating patients suffering from post-treatment Lyme disease syndrome (PTLDS) or chronic Lyme disease.
So, how exactly does PGLYRP1 work? PGLYRP1 is known to be a bactericidal protein expressed by innate immune cells, primarily neutrophils. It also is known that neutrophils play a critical role in killing and clearing spirochetes from infected tissues, particularly joints.7 Dr Gupta found that exposing B. burgdorferi to human PGLYRP1 in a test tube for 48 hours allowed the host protein to bind to the bacterial PG and subsequently kill the spirochetes. Of course, the $64,000 question is whether this killing effect can occur in a patient and, if so, could this lead to a novel approach to treating patients suffering from post-treatment Lyme disease syndrome (PTLDS) or chronic Lyme disease.
Finding more effective ways of treating this patient population is an overarching goal of GLA and one that takes on greater urgency considering the results of a study by scientists at GLA and Brown University that estimates more than two million people could suffer from PTLDS by years’ end. Additional excitement about the potential for a novel treatment option in the form of PGLYRP1 derives from the fact that PTLDS patients have received conventional standard-of-care antibiotics, such as Doxycycline, and yet continue to suffer from unremitting symptoms.
To read more Research/Science blogs, click here.
1https://www.cdc.gov/mmwr/volumes/67/wr/mm6717e1.htm?s_cid=mm6717e1_w
3https://journals.plos.org/plospathogens/article?id=10.1371/journal.ppat.1009030
4https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0093243
5https://pubmed.ncbi.nlm.nih.gov/29311239/
6https://pubmed.ncbi.nlm.nih.gov/31209025/
7https://pubmed.ncbi.nlm.nih.gov/21281805/

Timothy Sellati, P.h.D.
Former Chief Scientific Officer at Global Lyme Alliance
Timothy J. Sellati, PH.D. is Former Chief Scientific Officer at Global Lyme Alliance. As GLA’s Chief Scientific Officer, Dr. Sellati led GLA’s research initiatives to accelerate the development of more effective methods of diagnosis and treatment of Lyme and other tick-borne diseases.

-3.jpg)
-1.jpg)

-2.jpg)

