Important study identifies the Borrelia burgdorferi peptidoglycan as an immunogen likely to cause Lyme arthritis in some patients
by Mayla Hsu, Ph.D., Director of Research and Science, GLA
A longstanding mystery in Lyme disease is why some antibiotic-treated patients continue to suffer long-term symptoms, while others recover. It is possible that a hyperactive immune system in some patients causes inflammation-related tissue damage. Another possibility is that the bacteria, Borrelia burgdorferi, may still be alive in compartments of the body that are not readily accessible to antibiotics, and the bacteria’s continued replication causes ongoing symptoms.
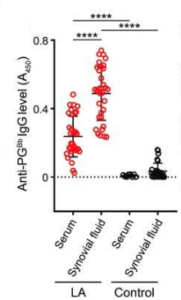
A recent article found that a component of the bacterial cell wall, peptidoglycan (PG), might be driving the persistent inflammation that causes Lyme arthritis even after antibiotic treatment. The authors, who include a GLA-funded researcher, found B. burgdorferi PG antibodies in the synovial fluid that surrounds the knees of Lyme arthritis patients, which is evidence of an ongoing immune response against bacteria or fragments of its cell wall. In contrast, they did not find significant antibodies against PGs of any other bacteria. A strength of this study is that they compared synovial fluid from Lyme arthritis patients with that of other types of non Lyme-related arthritis. Only Lyme arthritis patients had synovial fluid B. burgdorferi PG antibodies.
Blood was also collected from the same Lyme patients whose synovial fluid was studied. Here, they found that sera (serums) of Lyme arthritis patients also had PG antibodies, although the levels were much lower than in the synovial fluid. Moreover, Lyme arthritis patient sera had much higher levels of PG antibodies than any detected in control patient sera. These findings indicate a continued immune response against PG, particularly in the synovial joints, specifically against B. burgdorferi.
Another asset of this study was its analysis of patient samples both before and after antibiotic treatment. The authors designed a way to detect PG itself, not only antibodies, in synovial fluid and sera. Using this test, they could not find PG in control synovial fluid, nor in Lyme arthritis patient sera. However, it was present in 92% of Lyme arthritis synovial fluids, both before and after treatment with antibiotics; and this was true of patients who had been treated with oral as well as intravenous antibiotics. In addition, the amount of PG strongly correlated with the level of PG antibodies in the same synovial fluid samples. This finding suggests that the bacterial cell wall itself is present in the synovia of patients, and that its presence elicits antibodies against PG.
Do these results mean that B. burgdorferi is actively replicating in the patients in whom PG or PG antibodies were found? Testing for bacterial DNA was done on synovial fluid and blood, both before and after antibiotic treatment. Although both compartments were often positive for bacterial DNA before therapy, almost all samples became negative after therapy. This finding suggests that PG and PG antibodies persisted even after eliminating actively replicating B. burgdorferi at these sites, but it does not rule out bacterial replication elsewhere, and leaves open the question of how bacterial cell wall components could remain long after the bacterial DNA is no longer detectable.
This study also identified proinflammatory cytokines in synovial fluid, which play an important role in mediating immune responses. In vitro, the addition of purified B. burgdorferi PG to cultured peripheral blood mononuclear cells (PBMCs) caused increased levels of inflammatory cytokines like TNFα, IL1α, and IFNγ. These and others were also significantly elevated in the synovial fluid of Lyme arthritis patients, suggesting a possible mechanistic link between PG and inflammation. Finally, injection of purified PG in mice resulted in ankle swelling and inflammatory changes that are consistent with the established mouse model for Lyme arthritis.
In sum, these results make a strong case that B. burgdorferi PG may be responsible for driving inflammation and continued symptoms in post-antibiotic Lyme arthritis. Whether it is also implicated in promoting other long-term symptoms remains to be seen. These findings open up a new avenue of inquiry that may yield fruitful insights that will help us to better understand and care for people suffering from persistent symptoms.
Learn more about GLA’s research initiatives and accomplishments:
Research Report
Published Research Findings
Current Grantees
First Observational Study for Lyme Disease Treatment
Post-treatment Lyme: Two Million by 2020
Blog: Why Good Science is Crucial

GLA
Admin at GLA

-3.jpg)
-1.jpg)

-2.jpg)

